what two chemicals link up to produce energy?
Chapter 4: Introduction to How Cells Obtain Energy
4.1 Energy and Metabolism
Learning Objectives
By the end of this section, you volition be able to:
- Explain what metabolic pathways are
- State the commencement and second laws of thermodynamics
- Explain the deviation between kinetic and potential energy
- Draw endergonic and exergonic reactions
- Discuss how enzymes function every bit molecular catalysts
Lookout man a video nigh heterotrophs.
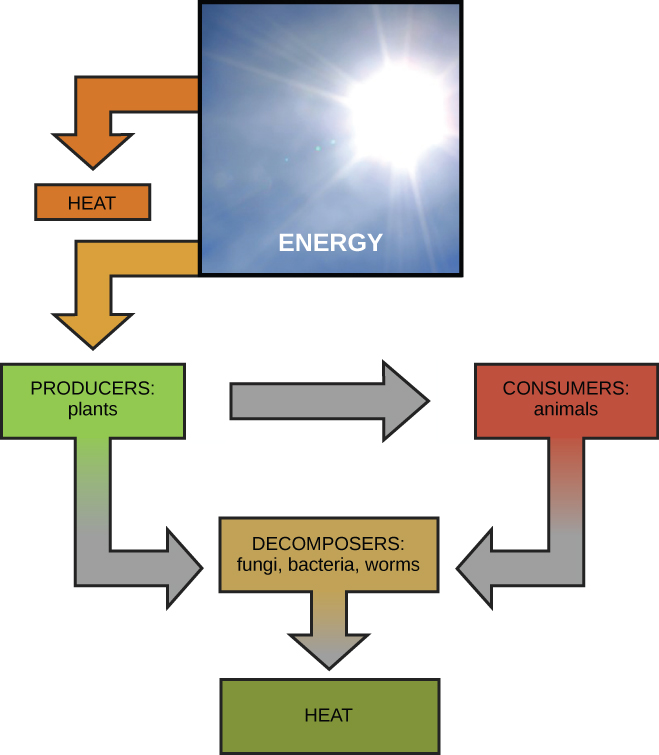
Scientists use the term bioenergetics to describe the concept of energy flow (Figure 4.2) through living systems, such as cells. Cellular processes such as the building and breaking downward of complex molecules occur through stepwise chemical reactions. Some of these chemic reactions are spontaneous and release energy, whereas others crave energy to proceed. Just equally living things must continually swallow food to replenish their energy supplies, cells must continually produce more energy to replenish that used by the many energy-requiring chemical reactions that constantly take place. Together, all of the chemical reactions that accept place inside cells, including those that eat or generate energy, are referred to as the cell's metabolism.
Metabolic Pathways
Consider the metabolism of sugar. This is a archetype example of i of the many cellular processes that use and produce energy. Living things consume sugars every bit a major energy source, considering sugar molecules have a bully deal of energy stored within their bonds. For the most part, photosynthesizing organisms like plants produce these sugars. During photosynthesis, plants use energy (originally from sunlight) to catechumen carbon dioxide gas (CO2) into saccharide molecules (like glucose: CsixH12Ovi). They swallow carbon dioxide and produce oxygen every bit a waste product product. This reaction is summarized every bit:
6CO2 + 6HtwoO + free energy ——-> Chalf dozenH12O6+ 6O2
Because this process involves synthesizing an energy-storing molecule, information technology requires energy input to continue. During the light reactions of photosynthesis, free energy is provided by a molecule called adenosine triphosphate (ATP), which is the primary energy currency of all cells. Just equally the dollar is used as currency to purchase appurtenances, cells use molecules of ATP as energy currency to perform immediate work. In contrast, energy-storage molecules such as glucose are consumed only to be cleaved down to utilise their free energy. The reaction that harvests the free energy of a sugar molecule in cells requiring oxygen to survive can be summarized by the opposite reaction to photosynthesis. In this reaction, oxygen is consumed and carbon dioxide is released as a waste matter product. The reaction is summarized every bit:
C6H12O6 + 6Oii ——> 6CO2 + 6HtwoO + free energy
Both of these reactions involve many steps.
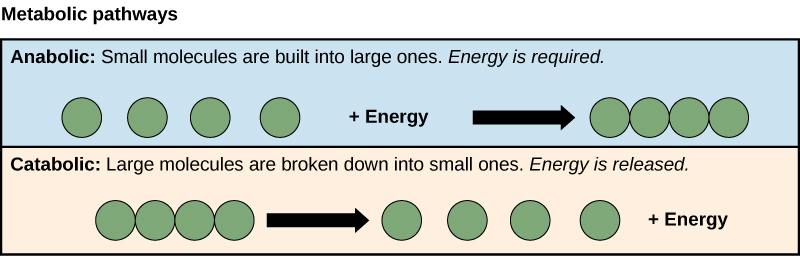
The processes of making and breaking downwardly sugar molecules illustrate two examples of metabolic pathways. A metabolic pathway is a series of chemic reactions that takes a starting molecule and modifies it, step-by-step, through a series of metabolic intermediates, eventually yielding a final product. In the instance of sugar metabolism, the commencement metabolic pathway synthesized saccharide from smaller molecules, and the other pathway broke sugar downwards into smaller molecules. These two contrary processes—the first requiring energy and the second producing free energy—are referred to every bit anabolic pathways (edifice polymers) and catabolic pathways (breaking down polymers into their monomers), respectively. Consequently, metabolism is composed of synthesis (anabolism) and deposition (catabolism) (Figure 4.3).
It is important to know that the chemic reactions of metabolic pathways practise not take place on their own. Each reaction step is facilitated, or catalyzed, by a protein called an enzyme. Enzymes are important for catalyzing all types of biological reactions—those that crave free energy equally well as those that release energy.
Energy
Thermodynamics refers to the study of energy and energy transfer involving concrete matter. The thing relevant to a detail case of energy transfer is chosen a system, and everything outside of that affair is called the environs. For example, when heating a pot of water on the stove, the organisation includes the stove, the pot, and the water. Free energy is transferred within the system (between the stove, pot, and water). There are two types of systems: open and closed. In an open up organization, energy can be exchanged with its environment. The stovetop system is open because rut can exist lost to the air. A closed system cannot exchange energy with its environment.
Biological organisms are open systems. Energy is exchanged between them and their surroundings as they employ energy from the sun to perform photosynthesis or consume free energy-storing molecules and release free energy to the surround past doing work and releasing heat. Similar all things in the concrete world, energy is subject to physical laws. The laws of thermodynamics govern the transfer of energy in and amidst all systems in the universe.
In full general, free energy is defined as the power to do piece of work, or to create some kind of change. Energy exists in different forms. For example, electrical energy, light energy, and heat energy are all different types of energy. To appreciate the way energy flows into and out of biological systems, it is important to empathise two of the physical laws that govern energy.
Thermodynamics
The start police of thermodynamics states that the total amount of energy in the universe is abiding and conserved. In other words, in that location has always been, and always will exist, exactly the same amount of energy in the universe. Energy exists in many dissimilar forms. Co-ordinate to the first law of thermodynamics, free energy may be transferred from place to place or transformed into different forms, simply it cannot be created or destroyed. The transfers and transformations of energy take place around us all the fourth dimension. Light bulbs transform electrical energy into calorie-free and oestrus energy. Gas stoves transform chemical free energy from natural gas into heat energy. Plants perform 1 of the nigh biologically useful energy transformations on world: that of converting the free energy of sunlight to chemic free energy stored within organic molecules (Figure four.2). Some examples of energy transformations are shown in Figure iv.4.
The challenge for all living organisms is to obtain energy from their surroundings in forms that they can transfer or transform into usable energy to do work. Living cells have evolved to come across this claiming. Chemical energy stored within organic molecules such equally sugars and fats is transferred and transformed through a series of cellular chemical reactions into energy within molecules of ATP. Energy in ATP molecules is hands accessible to practise work. Examples of the types of work that cells need to do include building complex molecules, transporting materials, powering the motion of cilia or flagella, and contracting muscle fibers to create movement.

A living cell's principal tasks of obtaining, transforming, and using energy to do work may seem simple. However, the second law of thermodynamics explains why these tasks are harder than they announced. All energy transfers and transformations are never completely efficient. In every energy transfer, some corporeality of free energy is lost in a form that is unusable. In virtually cases, this course is rut free energy. Thermodynamically, rut free energy is divers as the energy transferred from one system to another that is not work. For example, when a light bulb is turned on, some of the energy being converted from electrical free energy into light energy is lost as rut free energy. Likewise, some energy is lost as estrus free energy during cellular metabolic reactions.
An important concept in physical systems is that of gild and disorder. The more than energy that is lost by a organisation to its surround, the less ordered and more than random the system is. Scientists refer to the measure out of randomness or disorder within a system as entropy. High entropy means high disorder and low energy. Molecules and chemical reactions accept varying entropy as well. For case, entropy increases as molecules at a high concentration in one place diffuse and spread out. The second law of thermodynamics says that energy will always exist lost as estrus in energy transfers or transformations.
Living things are highly ordered, requiring constant energy input to exist maintained in a land of depression entropy.
Potential and Kinetic Free energy
When an object is in motion, at that place is energy associated with that object. Retrieve of a wrecking brawl. Even a ho-hum-moving wrecking ball tin do a great deal of damage to other objects. Energy associated with objects in move is called kinetic energy (Effigy four.five). A speeding bullet, a walking person, and the rapid movement of molecules in the air (which produces heat) all accept kinetic energy.
Now what if that same motionless wrecking ball is lifted 2 stories above ground with a crane? If the suspended wrecking ball is unmoving, is there energy associated with it? The answer is yes. The energy that was required to lift the wrecking ball did non disappear, only is at present stored in the wrecking ball by virtue of its position and the force of gravity acting on it. This blazon of energy is called potential energy (Figure 4.5). If the ball were to autumn, the potential energy would be transformed into kinetic energy until all of the potential energy was exhausted when the ball rested on the ground. Wrecking balls as well swing like a pendulum; through the swing, there is a constant change of potential energy (highest at the height of the swing) to kinetic energy (highest at the bottom of the swing). Other examples of potential free energy include the free energy of water held behind a dam or a person virtually to skydive out of an airplane.

Potential energy is not but associated with the location of matter, merely also with the structure of matter. Fifty-fifty a jump on the ground has potential energy if information technology is compressed; then does a prophylactic band that is pulled taut. On a molecular level, the bonds that hold the atoms of molecules together be in a particular structure that has potential energy. Remember that anabolic cellular pathways require energy to synthesize complex molecules from simpler ones and catabolic pathways release energy when complex molecules are broken downwards. The fact that energy tin be released by the breakdown of certain chemical bonds implies that those bonds take potential free energy. In fact, there is potential energy stored inside the bonds of all the nutrient molecules we consume, which is eventually harnessed for use. This is because these bonds can release energy when broken. The type of potential free energy that exists within chemical bonds, and is released when those bonds are cleaved, is called chemical energy. Chemic energy is responsible for providing living cells with free energy from food. The release of energy occurs when the molecular bonds within food molecules are cleaved.
Sentinel a video about kilocalories.
Concept in Action

Visit the site and select "Pendulum" from the "Work and Energy" menu to run across the shifting kinetic and potential energy of a pendulum in motility.
Free and Activation Energy
After learning that chemical reactions release free energy when energy-storing bonds are broken, an important next question is the following: How is the free energy associated with these chemical reactions quantified and expressed? How can the free energy released from i reaction be compared to that of some other reaction? A measurement of complimentary energy is used to quantify these energy transfers. Call back that according to the second law of thermodynamics, all energy transfers involve the loss of some corporeality of energy in an unusable form such as heat. Free free energy specifically refers to the energy associated with a chemical reaction that is available afterward the losses are accounted for. In other words, free energy is usable energy, or energy that is bachelor to do piece of work.
If energy is released during a chemical reaction, then the change in free energy, signified equally ∆G (delta G) volition be a negative number. A negative change in free energy also means that the products of the reaction have less gratis energy than the reactants, because they release some gratis energy during the reaction. Reactions that have a negative alter in energy and consequently release energy are called exergonic reactions. Think: exergonic means energy is exiting the system. These reactions are as well referred to every bit spontaneous reactions, and their products have less stored energy than the reactants. An important distinction must be fatigued between the term spontaneous and the idea of a chemical reaction occurring immediately. Contrary to the everyday employ of the term, a spontaneous reaction is not one that suddenly or quickly occurs. The rusting of iron is an example of a spontaneous reaction that occurs slowly, piddling by little, over fourth dimension.
If a chemical reaction absorbs energy rather than releases energy on residual, then the ∆Thousand for that reaction will be a positive value. In this case, the products accept more than free energy than the reactants. Thus, the products of these reactions can exist thought of as energy-storing molecules. These chemical reactions are called endergonic reactions and they are non-spontaneous. An endergonic reaction will not have place on its own without the addition of gratis energy.

Await at each of the processes shown and make up one's mind if it is endergonic or exergonic.
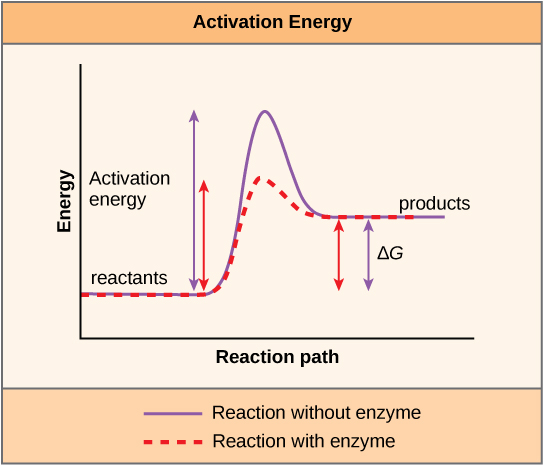
There is another important concept that must be considered regarding endergonic and exergonic reactions. Exergonic reactions require a modest corporeality of energy input to get going, before they can continue with their energy-releasing steps. These reactions have a net release of energy, merely however require some energy input in the beginning. This modest amount of energy input necessary for all chemical reactions to occur is chosen the activation energy.
Concept in Action

Watch an animation of the motion from energy to transition land of the reaction.
Enzymes
A substance that helps a chemical reaction to occur is called a catalyst, and the molecules that catalyze biochemical reactions are called enzymes. Most enzymes are proteins and perform the critical chore of lowering the activation energies of chemical reactions inside the cell. Most of the reactions critical to a living cell happen likewise slowly at normal temperatures to be of whatever use to the cell. Without enzymes to speed up these reactions, life could not persist. Enzymes practise this by bounden to the reactant molecules and holding them in such a style equally to make the chemic bail-breaking and -forming processes have identify more easily. Information technology is important to call up that enzymes do not change whether a reaction is exergonic (spontaneous) or endergonic. This is because they practise not modify the costless energy of the reactants or products. They only reduce the activation energy required for the reaction to become forrard (Effigy 4.seven). In addition, an enzyme itself is unchanged by the reaction information technology catalyzes. Once 1 reaction has been catalyzed, the enzyme is able to participate in other reactions.
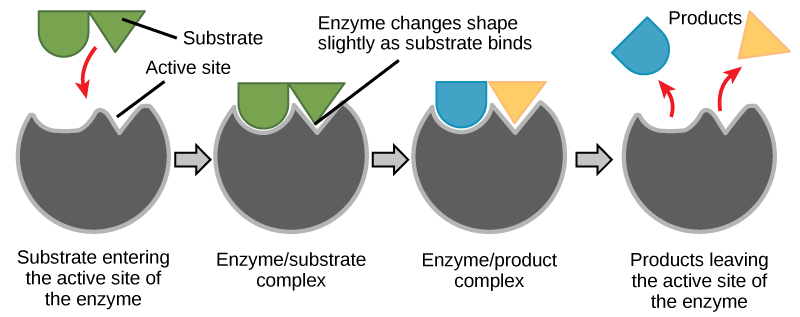
The chemical reactants to which an enzyme binds are called the enzyme's substrates. At that place may exist one or more than substrates, depending on the particular chemical reaction. In some reactions, a unmarried reactant substrate is broken down into multiple products. In others, ii substrates may come together to create one larger molecule. Two reactants might too enter a reaction and both become modified, but they leave the reaction equally two products. The location within the enzyme where the substrate binds is called the enzyme's active site. The active site is where the "action" happens. Since enzymes are proteins, there is a unique combination of amino acid side chains inside the active site. Each side concatenation is characterized by different properties. They tin be big or small, weakly acidic or bones, hydrophilic or hydrophobic, positively or negatively charged, or neutral. The unique combination of side bondage creates a very specific chemical environment inside the agile site. This specific environment is suited to demark to one specific chemical substrate (or substrates).
Active sites are subject to influences of the local environment. Increasing the environmental temperature generally increases reaction rates, enzyme-catalyzed or otherwise. However, temperatures outside of an optimal range reduce the rate at which an enzyme catalyzes a reaction. Hot temperatures will eventually cause enzymes to denature, an irreversible change in the three-dimensional shape and therefore the role of the enzyme. Enzymes are also suited to function best within a certain pH and common salt concentration range, and, every bit with temperature, extreme pH, and table salt concentrations can crusade enzymes to denature.
For many years, scientists thought that enzyme-substrate binding took place in a simple "lock and key" fashion. This model asserted that the enzyme and substrate fit together perfectly in ane instantaneous step. However, current research supports a model chosen induced fit (Figure 4.8). The induced-fit model expands on the lock-and-key model by describing a more dynamic binding between enzyme and substrate. As the enzyme and substrate come together, their interaction causes a mild shift in the enzyme'south structure that forms an ideal binding arrangement betwixt enzyme and substrate.
Concept in Action

View an animation of induced fit.
When an enzyme binds its substrate, an enzyme-substrate complex is formed. This complex lowers the activation energy of the reaction and promotes its rapid progression in i of multiple possible ways. On a basic level, enzymes promote chemical reactions that involve more than one substrate by bringing the substrates together in an optimal orientation for reaction. Another manner in which enzymes promote the reaction of their substrates is by creating an optimal environment within the active site for the reaction to occur. The chemical properties that sally from the particular arrangement of amino acid R groups within an active site create the perfect environment for an enzyme's specific substrates to react.
The enzyme-substrate circuitous can also lower activation energy by compromising the bond structure so that it is easier to suspension. Finally, enzymes can also lower activation energies by taking part in the chemical reaction itself. In these cases, it is of import to remember that the enzyme volition always render to its original state past the completion of the reaction. I of the hallmark properties of enzymes is that they remain ultimately unchanged by the reactions they catalyze. Subsequently an enzyme has catalyzed a reaction, it releases its product(s) and tin catalyze a new reaction.
It would seem ideal to have a scenario in which all of an organism's enzymes existed in arable supply and functioned optimally under all cellular weather condition, in all cells, at all times. However, a multifariousness of mechanisms ensures that this does not happen. Cellular needs and atmospheric condition constantly vary from jail cell to cell, and change inside individual cells over time. The required enzymes of stomach cells differ from those of fat storage cells, pare cells, claret cells, and nerve cells. Furthermore, a digestive organ prison cell works much harder to procedure and break down nutrients during the time that closely follows a repast compared with many hours after a repast. As these cellular demands and weather condition vary, then must the amounts and functionality of dissimilar enzymes.
Since the rates of biochemical reactions are controlled by activation energy, and enzymes lower and decide activation energies for chemic reactions, the relative amounts and functioning of the variety of enzymes within a prison cell ultimately determine which reactions will go along and at what rates. This determination is tightly controlled in cells. In certain cellular environments, enzyme activeness is partly controlled by environmental factors like pH, temperature, salt concentration, and, in some cases, cofactors or coenzymes.
Enzymes tin also be regulated in means that either promote or reduce enzyme activity. There are many kinds of molecules that inhibit or promote enzyme function, and various mechanisms by which they do and so. In some cases of enzyme inhibition, an inhibitor molecule is similar plenty to a substrate that it can demark to the active site and simply block the substrate from binding. When this happens, the enzyme is inhibited through competitive inhibition, because an inhibitor molecule competes with the substrate for binding to the active site.
On the other hand, in noncompetitive inhibition, an inhibitor molecule binds to the enzyme in a location other than the active site, called an allosteric site, but still manages to block substrate binding to the active site. Some inhibitor molecules bind to enzymes in a location where their binding induces a conformational modify that reduces the affinity of the enzyme for its substrate. This type of inhibition is called allosteric inhibition (Figure 4.9). Nearly allosterically regulated enzymes are fabricated upward of more than than one polypeptide, meaning that they accept more than one protein subunit. When an allosteric inhibitor binds to a region on an enzyme, all active sites on the protein subunits are changed slightly such that they bind their substrates with less efficiency. There are allosteric activators too every bit inhibitors. Allosteric activators bind to locations on an enzyme abroad from the agile site, inducing a conformational change that increases the affinity of the enzyme'south agile site(s) for its substrate(s) (Figure four.9).
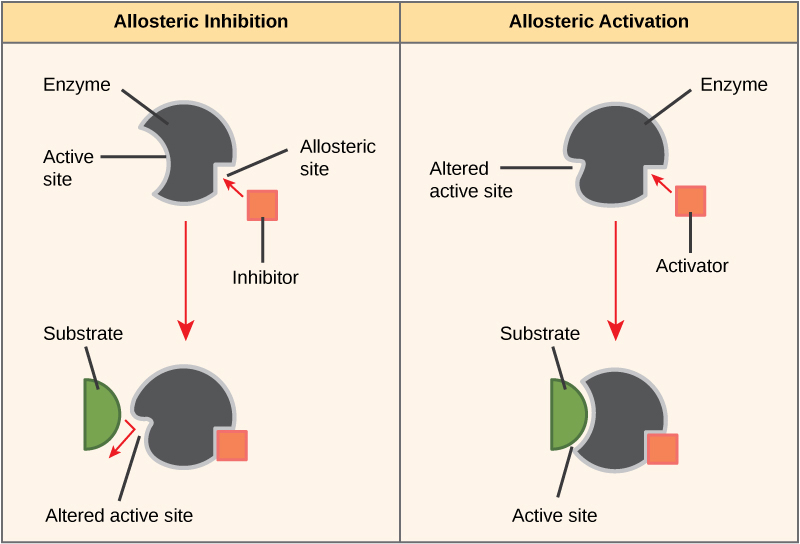
Through the Indigenous Lens
Plants cannot run or hide from their predators and have evolved many strategies to deter those who would eat them. Think of thorns, irritants and secondary metabolites: these are compounds that do non directly help the establish grow, but are made specifically to continue predators away. Secondary metabolites are the most mutual way plants deter predators. Some examples of secondary metabolites are atropine, nicotine, THC and caffeine. Humans take found these secondary metabolite compounds a rich source of materials for medicines. It is estimated that ninety% of the drugs in the modern pharmacy have their "roots" in these secondary metabolites.
Get-go peoples herbal treatments revealed these secondary metabolites to the world. For example, Indigenous peoples accept long used the bark of willow shrubs and alder trees for a tea, tonic or poultice to reduce inflammation. Yous volition learn more about the inflammation response by the immune arrangement in chapter xi.

Both willow and alder bark incorporate the compound salicin. Most of us have this compound in our medicine cupboard in the form of salicylic acid or aspirin. Aspirin has been proved to reduce pain and inflammation, and in one case in our cells salicin converts to salicylic acid.
So how does it work? Salicin or aspirin acts every bit an enzyme inhibitor. In the inflammatory response two enzymes, COX1 and COX2 are key to this process. Salicin or aspirin specifically modifies an amino acid (serine) in the active site of these ii related enzymes. This modification of the active sites does not permit the normal substrate to bind so the inflammatory process is disrupted. As y'all have read in this chapter, this makes it competitive enzyme inhibitor.
Pharmaceutical Drug Programmer

Enzymes are key components of metabolic pathways. Understanding how enzymes work and how they can be regulated are key principles behind the development of many of the pharmaceutical drugs on the market place today. Biologists working in this field collaborate with other scientists to pattern drugs (Figure four.11).
Consider statins for case—statins is the name given to one form of drugs that can reduce cholesterol levels. These compounds are inhibitors of the enzyme HMG-CoA reductase, which is the enzyme that synthesizes cholesterol from lipids in the body. By inhibiting this enzyme, the level of cholesterol synthesized in the body can be reduced. Similarly, acetaminophen, popularly marketed under the brand name Tylenol, is an inhibitor of the enzyme cyclooxygenase. While it is used to provide relief from fever and inflammation (pain), its mechanism of activity is withal not completely understood.
How are drugs discovered? 1 of the biggest challenges in drug discovery is identifying a drug target. A drug target is a molecule that is literally the target of the drug. In the instance of statins, HMG-CoA reductase is the drug target. Drug targets are identified through painstaking research in the laboratory. Identifying the target alone is not enough; scientists as well need to know how the target acts inside the cell and which reactions get awry in the instance of disease. In one case the target and the pathway are identified, then the actual procedure of drug blueprint begins. In this stage, chemists and biologists work together to pattern and synthesize molecules that can block or activate a particular reaction. However, this is only the beginning: If and when a drug epitome is successful in performing its function, then information technology is subjected to many tests from in vitro experiments to clinical trials before it tin get blessing from the U.S. Food and Drug Assistants to exist on the market.
Many enzymes practice not work optimally, or even at all, unless jump to other specific not-poly peptide helper molecules. They may bond either temporarily through ionic or hydrogen bonds, or permanently through stronger covalent bonds. Binding to these molecules promotes optimal shape and part of their respective enzymes. Ii examples of these types of helper molecules are cofactors and coenzymes. Cofactors are inorganic ions such as ions of iron and magnesium. Coenzymes are organic helper molecules, those with a basic diminutive structure fabricated up of carbon and hydrogen. Like enzymes, these molecules participate in reactions without being changed themselves and are ultimately recycled and reused. Vitamins are the source of coenzymes. Some vitamins are the precursors of coenzymes and others act directly every bit coenzymes. Vitamin C is a straight coenzyme for multiple enzymes that take office in building the important connective tissue, collagen. Therefore, enzyme role is, in part, regulated by the abundance of various cofactors and coenzymes, which may be supplied by an organism'due south nutrition or, in some cases, produced by the organism.
Feedback Inhibition in Metabolic Pathways
Molecules can regulate enzyme office in many ways. The major question remains, however: What are these molecules and where do they come from? Some are cofactors and coenzymes, as you have learned. What other molecules in the jail cell provide enzymatic regulation such every bit allosteric modulation, and competitive and non-competitive inhibition? Peradventure the most relevant sources of regulatory molecules, with respect to enzymatic cellular metabolism, are the products of the cellular metabolic reactions themselves. In a near efficient and elegant way, cells take evolved to use the products of their own reactions for feedback inhibition of enzyme action. Feedback inhibition involves the use of a reaction product to regulate its own further production (Figure 4.12). The cell responds to an abundance of the products by slowing down product during anabolic or catabolic reactions. Such reaction products may inhibit the enzymes that catalyzed their production through the mechanisms described to a higher place.
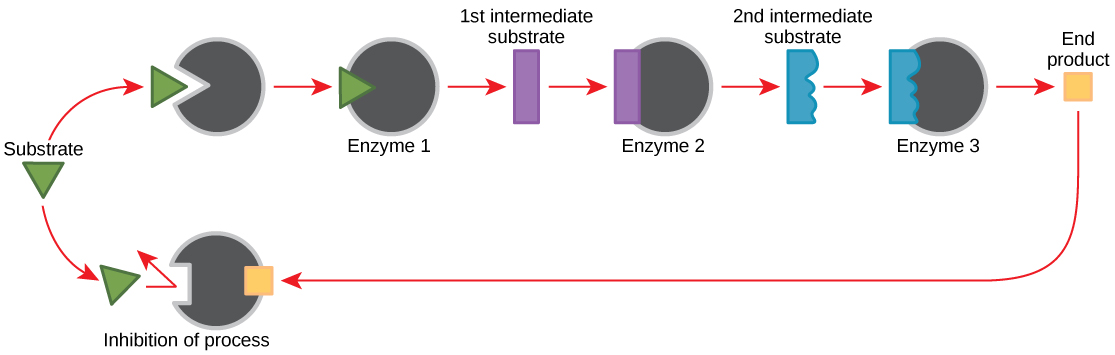
The production of both amino acids and nucleotides is controlled through feedback inhibition. Additionally, ATP is an allosteric regulator of some of the enzymes involved in the catabolic breakdown of sugar, the process that creates ATP. In this way, when ATP is in abundant supply, the cell tin can prevent the production of ATP. On the other hand, ADP serves every bit a positive allosteric regulator (an allosteric activator) for some of the same enzymes that are inhibited past ATP. Thus, when relative levels of ADP are high compared to ATP, the cell is triggered to produce more than ATP through sugar catabolism.
Section Summary
Cells perform the functions of life through various chemical reactions. A cell'due south metabolism refers to the combination of chemical reactions that take place within it. Catabolic reactions interruption down complex chemicals into simpler ones and are associated with energy release. Anabolic processes build complex molecules out of simpler ones and require energy.
In studying free energy, the term system refers to the matter and surround involved in energy transfers. Entropy is a measure of the disorder of a organisation. The physical laws that describe the transfer of energy are the laws of thermodynamics. The offset law states that the full corporeality of energy in the universe is constant. The 2nd constabulary of thermodynamics states that every energy transfer involves some loss of energy in an unusable form, such as oestrus energy. Energy comes in different forms: kinetic, potential, and gratuitous. The change in free energy of a reaction can exist negative (releases energy, exergonic) or positive (consumes energy, endergonic). All reactions require an initial input of energy to proceed, called the activation energy.
Enzymes are chemical catalysts that speed upwardly chemical reactions by lowering their activation free energy. Enzymes have an active site with a unique chemical environs that fits item chemical reactants for that enzyme, called substrates. Enzymes and substrates are idea to bind according to an induced-fit model. Enzyme action is regulated to conserve resource and answer optimally to the surroundings.
Glossary
activation energy: the amount of initial free energy necessary for reactions to occur
active site: a specific region on the enzyme where the substrate binds
allosteric inhibition: the machinery for inhibiting enzyme action in which a regulatory molecule binds to a 2nd site (not the agile site) and initiates a conformation change in the agile site, preventing binding with the substrate
anabolic: describes the pathway that requires a net energy input to synthesize complex molecules from simpler ones
bioenergetics: the concept of energy flow through living systems
catabolic: describes the pathway in which complex molecules are broken down into simpler ones, yielding energy as an additional production of the reaction
competitive inhibition: a general mechanism of enzyme action regulation in which a molecule other than the enzyme'due south substrate is able to bind the active site and prevent the substrate itself from bounden, thus inhibiting the overall rate of reaction for the enzyme
endergonic: describes a chemical reaction that results in products that store more chemical potential energy than the reactants
enzyme: a molecule that catalyzes a biochemical reaction
exergonic: describes a chemic reaction that results in products with less chemical potential energy than the reactants, plus the release of free free energy
feedback inhibition: a mechanism of enzyme activity regulation in which the product of a reaction or the last production of a series of sequential reactions inhibits an enzyme for an earlier step in the reaction series
heat free energy: the energy transferred from one arrangement to another that is non piece of work
kinetic energy: the type of free energy associated with objects in motility
metabolism: all the chemical reactions that take place inside cells, including those that use energy and those that release energy
noncompetitive inhibition: a full general machinery of enzyme activity regulation in which a regulatory molecule binds to a site other than the active site and prevents the active site from binding the substrate; thus, the inhibitor molecule does non compete with the substrate for the active site; allosteric inhibition is a form of noncompetitive inhibition
potential energy: the type of energy that refers to the potential to do piece of work
substrate: a molecule on which the enzyme acts
thermodynamics: the science of the relationships between heat, free energy, and piece of work
Source: https://opentextbc.ca/biology/chapter/4-1-energy-and-metabolism/
0 Response to "what two chemicals link up to produce energy?"
Post a Comment